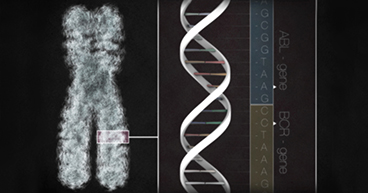
Our DNA makes us who we are. It determines our hair and eye color and, to some extent, our physical abilities and talents. But the double helix has also made an indelible impact beyond our own bodies. These molecular building blocks have played a vital role in the evolution of health care, helping us better understand diseases like cancer, and the scientific community’s efforts to treat them.
There’s lots to celebrate in these achievements, and today is the day set aside to do just that: recognize and applaud the science, knowledge and perseverance inherent in the quest to understand what our DNA looks like and how it works.
This National DNA Day, we celebrate two milestones that helped unlock the mysteries of DNA, leading to a cascade of scientific discoveries and new treatments for diseases like cancer:
- In 1953, molecular biologists James Watson and Francis Crick discovered the iconic double-helix structure of the DNA strand. This discovery led to information about how the rungs on the DNA ladder are organized, how the strands can replicate themselves and how they store and transmit information.
- In 2003, scientists completed the Human Genome Project, which mapped the genetic material inside human cells. The project help to crystallize our understanding of the structure of DNA, genes and chromosomes. It has also led to a better understanding of how mutations in our genes can lead to disease, including cancer.
Below are five ways these discoveries have helped scientists and doctors better understand cancer and how to treat it.
Gene mutations that cause cancer
Scientists have known for decades that cancer was caused by changes in human DNA. The Human Genome Project not only confirmed those theories; it helped identify specific mutations and improve our understanding of why they may lead to cancer. Scientists also have been able to classify types of genes that are found in many cancers:
Proto-oncogenes: These genes regulate how cells grow. When proto-oncogenes mutate, they become oncogenes—genes that can cause cancer. Examples of common oncogenes include:
- EGFR, found in pancreatic, head and neck and colorectal cancers
- HER2, found in breast cancer
Tumor suppressor genes: These genes regulate when cells stop growing. They also shut down defective cells or those that grow too fast and sometimes repair damage in DNA. When tumor suppressor cells mutate, damaged cells may be allowed to grow and potentially form cancers. Common tumor suppressor cells found in cancers include:
- TP53 or P53, found in about half of all cancers
- BRCA1 and BRCA2, found in about 10 percent of breast cancers and in reproductive cancers, pancreatic cancer and other cancers
“Following the mapping of the human genome, investigators were able to define specific molecular defects in individual patients linked to the growth of that patient’s cancer. They subsequently developed strategies to target that abnormality for the purpose of interfering with the progression of the cancer,” says Maurie Markman, MD, President of Medicine & Science at Cancer Treatment Centers of America® (CTCA).
Targeted therapy
Armed with the knowledge of how to identify specific gene mutations in cancers, scientists would soon take the next step: developing drugs designed to target those specific features in tumors. That’s where targeted therapy drugs come in. They’re tailored to attack certain mutations in cancer cells and kill the cells or help other treatments work better. The drugs often target cancer cells’ ability to grow unchecked, but some are designed to stop certain survival techniques cancers use to adapt to threats, such as creating new blood supplies.
Still, identifying gene mutations in tumors does not guarantee a targeted therapy drug can be developed for that mutation, or that the drug will help slow a tumor’s growth, because:
- Tumors often have multiple mutations, making it difficult to identify those that are driving a tumor's growth.
- Driver mutations often mutate themselves, or allow other mutations in the tumor to take control of the growth, creating a moving target for doctors.
- Targeting certain genes may cause serious side effects.
“Much has been learned regarding the complexity of the genome, including the fact that individual genes control a number of vital normal processes, even if they’re also somehow related to cancer growth,” Dr. Markman says. “So, targeting a gene to interfere with its function has the potential downside of also interfering with normal activities of cells and body organs.”
Hereditary causes of cancer
Genes that determine hair and eye color, body type and other physical characteristics are passed down through our genes from one generation to the next. Unfortunately, some gene mutations are inherited in the same way. Mapping the human genome has helped doctors identify some hereditary mutations that may lead to cancer. These gene disorders, called hereditary cancer syndromes, are associated with about 10 percent of all cancers.
Being born with a cancer syndrome doesn’t mean you’ll get cancer. But it may mean you’re at higher risk. It also may help empower you to make decisions about whether to seek interventions. For example, patients with syndromes that increase breast cancer risks may decide on increased screenings to help detect cancer, or they may opt for aggressive prophylactic surgeries to help prevent it.
Learn more about hereditary cancer syndromes.
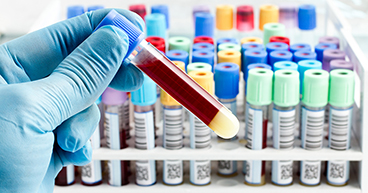
Liquid biopsies
Many cancer diagnoses require a biopsy to look for cancer cells and help determine the stage of the disease. Most biopsies are surgical procedures that remove a piece of a tumor that is then sent to a lab for examination. But what if doctors could perform biopsies without surgery?
Mapping the genome has led the way for liquid biopsies, which are tests using blood, urine, stool or other substances that may contain mutated cancer DNA. Several liquid biopsies already have been approved by the U.S. Food and Drug Administration to help detect gene mutations that may lead to some forms of lung cancer or colorectal cancer. They also may be used to measure cancer progression to determine if a treatment regimen is slowing the cancer’s growth. Liquid biopsies may not replace most traditional surgical biopsies, but they give doctors another option in their diagnostic toolbox.
“Liquid biopsies hold great promise as a strategy to more easily follow the natural course of molecular changes (for example, the development of drug resistance) within a cancer and subsequently permit changes in treatment strategies based on those findings,” Dr. Markman says.
Learn more about liquid biopsies.
CRISPR
Now that scientists know what DNA is supposed to look like and can identify errors or mutations that cause diseases such as cancer, scientists have pondered whether there’s a way to repair the errors in the DNA itself. Enter CRISPR—which stands for “clustered regularly interspaced short palindromic repeats.” In theory, CRISPR-Cas9, as it’s formally known, would help scientists find a faulty stretch of DNA, unravel it and match it to a strand of compatible RNA (ribose nucleic acid). Once rewoven, the strand would be repaired. CRISPR may also be used to re-wire cells to help them respond better to treatments for disease. Scientists at the University of Pennsylvania are conducting a clinical trial that uses CRISPR to edit patients’ T-cells to help them identify and attack cancer cells.
But CRISPR, like our understanding of the human genome, is in its infancy. Scientists are still trying to determine if the gene-editing tool can be applied practically and safely. For instance, could the repairing sequence in one stretch of DNA cause damage to a segment elsewhere in the strand? Even with so much unknown, CRISPR’s potential is believed to be so promising that it’s now being researched for use on diseases such as sickle cell anemia, cystic fibrosis and cancer.
“CRISPR is potentially an incredibly powerful strategy to relatively precisely change the molecular makeup of individual human cells,” Dr. Markman says. “There is a truly impressive opportunity for CRISPR to have a major favorable impact within a number of domains in clinical medicine, but its potential role in cancer care remains to be defined.”